iNCOVACC- Could this New Tool be the Answer to COVID?

The iNCOVACC has been received emergency use approval as a heterologous booster shot for individuals above 18 years of age. This move comes 3 months after emergency use approval of the nasal vaccine against COVID-19 for primary vaccination of adults.
This topic of “iNCOVACC- Could this New Tool be the Answer to COVID?” is important from the perspective of the UPSC IAS Examination, which falls under General Studies Portion.
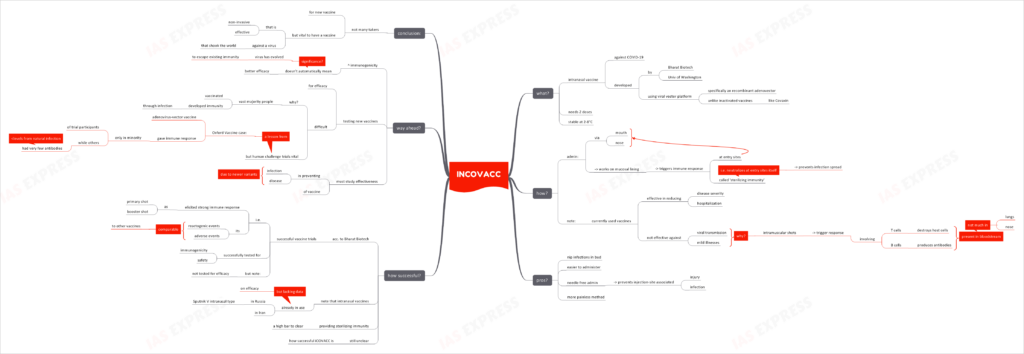
What is iNCOVACC?
- iNCOVACC is an intranasal vaccine against COVID-19.
- It was developed by Bharat Biotech, in association with the University of Washington.
- Unlike inactivated vaccines like Covaxin, the iNCOVACC has been developed using a viral vector platform. A recombinant adenovector has been used as the vector.
- It is a 2-shot vaccine.
- The vaccine is stable at 2-8°C.
How does it work?
- The vaccine is administered through the mouth or the nose.
- It works on the mucosal lining and prompts an immune response at the sites where the virus usually enters the body.
- The vaccine would thus have a sterilizing immunity effect i.e. prevents the virus at the entry sites itself to stop the spread of infection.
- The vaccines that are currently in use are effective in reducing disease severity and hospitalization. However, they aren’t capable of stopping the viral transmission or mild illnesses.
- This is because these vaccines are intramuscular shots i.e. introduced into the muscles.
- Such shots trigger an immune response involving:
- T cells: help destroy infected host cells
- B cells: produce antibodies to neutralize viruses
- While these immune cells are present in the bloodstream, they don’t occur in high enough concentrations in the nose or lungs. Hence, such intramuscular vaccines can’t protect individuals from getting ill.
What are the pros over other vaccines?
- Unlike the other vaccines, they can prevent SARS CoV 2 infection at the points of entry itself, thus nipping the infection in the bud.
- They are easier to administer.
- Needle-free administration implies elimination of injection-site injury and infection risks.
- It is also a more painless method of vaccination.
How successful are these vaccines?
- Bharat Biotech announced that the vaccine trials have been successful.
- The vaccine elicited strong immune response upon use as both- primary shot and also as a booster shot.
- The trials showed that reactogenic events and adverse events were comparable to those of other vaccines in use.
- However, while it was tested for immunogenicity and safety, it wasn’t tested for efficacy.
- Note that Russia had already deployed its intranasal version of Sputnik V and Iran has been using intranasal vaccines too. However, very little data is available on these vaccine’s efficacy.
- Expecting vaccines to provide sterilizing immunity is a high bar to clear and it is still unclear how successful these intranasal vaccines will be.
What is the way ahead?
- High immunogenicity doesn’t automatically mean better efficacy. This is especially significant given how the virus has evolved to increasingly evade existing immunity.
- Unlike in case of vaccines tested earlier in the pandemic, the newer vaccines would face challenges in efficacy evaluation as a vast majority of the people have already been vaccinated/ have developed immunity naturally through an infection.
- However, human challenge studies are vital before the vaccine can be widely deployed against the SARS CoV 2. This lesson was learnt from the Oxford vaccine’s case- the adenovirus-vector vaccine triggered antigen-specific mucosal antibody responses in only a minority of the trial participants. In other cases, the antibodies produced were much lower than the levels resulting from natural infection.
- It is important to study the vaccine’s effectiveness in preventing infections and disease due to the newer variants.
Conclusion:
Though there isn’t as many takers for COVID vaccine as in the initial stages, having a non-invasive but effective vaccine against the virus that shook the world is a high priority.
Practice Question for Mains:
What is iNCOVACC? How can it help in the fight against COVID-19? (250 words)

