[Newsbits] 03.08.2023
Fitch Downgrades USA
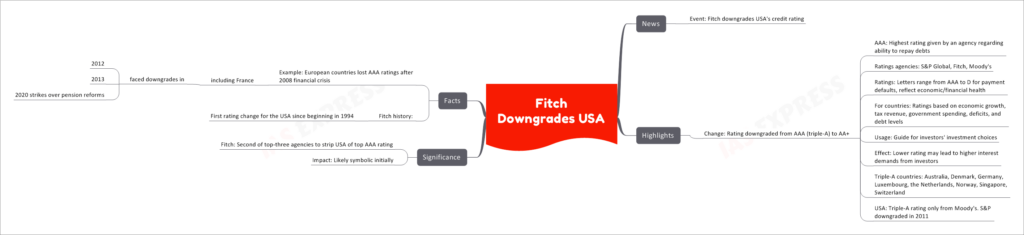
In recent news, Fitch has downgraded the credit rating of the USA from AAA (triple-A) to AA+.
Understanding Credit Ratings
Credit ratings are given by rating agencies like S&P Global, Fitch, and Moody's to assess the ability of an entity, such as a country or a company, to repay its debts. These ratings are represented by letter grades, ranging from AAA (highest rating) to D (indicating payment default). They reflect the economic and financial health of the entity and are based on various factors, including economic growth, tax revenue, government spending, deficits, and debt levels.
Investors often use these credit ratings as a guide for making investment choices. A higher credit rating usually indicates lower risk, while a lower rating may lead to higher interest demands from investors.
Fitch's Downgrade of USA's Credit Rating
The downgrade from AAA to AA+ is significant as it is the second of the top-three rating agencies to strip the USA of its top credit rating. Previously, S&P downgraded the USA in 2011.
Significance and Impact
The downgrade's immediate impact may be largely symbolic, but it could have implications in the long run. Historically, other countries, including European countries like France, lost their AAA ratings after the 2008 financial crisis and faced subsequent downgrades.
Facts and Examples
- Fitch's rating change is the first for the USA since the agency began rating the country in 1994.
- Currently, other countries with a AAA rating include Australia, Denmark, Germany, Luxembourg, the Netherlands, Norway, Singapore, and Switzerland.
- In the past, European countries like France experienced downgrades in their credit ratings during the 2012 and 2013 periods, as well as during strikes over pension reform in 2020.
Conclusion
The downgrade of the USA's credit rating by Fitch from AAA to AA+ marks a notable development and warrants attention from investors and policymakers alike. While the immediate impact may be limited, it signals potential concerns about the country's economic and financial outlook. As credit ratings influence borrowing costs and investor confidence, close monitoring of economic indicators and fiscal policies will be essential for the USA to maintain its creditworthiness in the global market.
China’s Plan to Limit Smartphone Usage
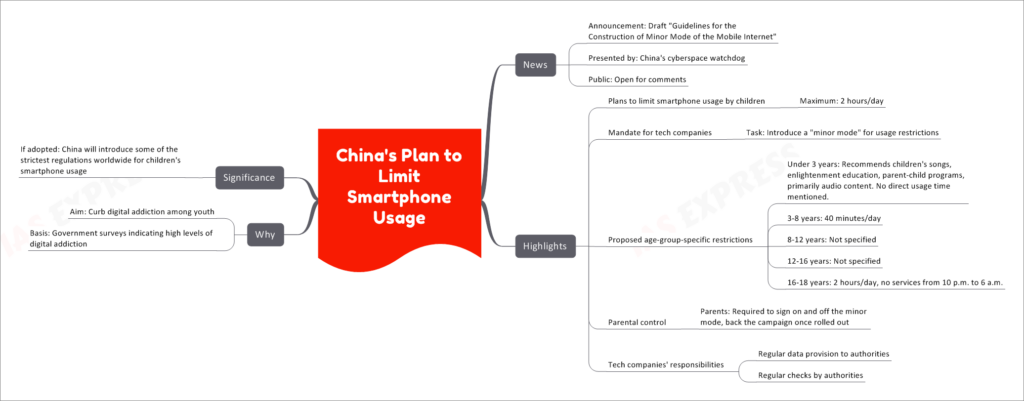
China's cyberspace watchdog has recently presented the draft "Guidelines for the Construction of Minor Mode of the Mobile Internet," aiming to limit smartphone usage among children and address digital addiction concerns. The draft is open for public comments and proposes age-group-specific restrictions for children's smartphone usage.
Highlights of the Guidelines
The draft guidelines include the following key points:
- Limiting Smartphone Usage by Children: The guidelines propose a maximum of 2 hours per day for children's smartphone usage.
- Introducing "Minor Mode" by Tech Companies: Tech companies will be mandated to introduce a "minor mode" with usage restrictions for children.
- Age-Group-Specific Restrictions: The proposed restrictions vary based on age groups, with recommended usage times as follows:
- Under 3 years: Recommends children's songs, enlightenment education, parent-child programs, primarily audio content. No direct usage time mentioned.
- 3-8 years: 40 minutes per day.
- 8-12 years: Not specified.
- 12-16 years: Not specified.
- 16-18 years: 2 hours per day, with no services available from 10 p.m. to 6 a.m.
- Parental Control: Parents will be required to sign on and off the minor mode, and they are expected to back the campaign once it is rolled out.
- Tech Companies' Responsibilities: Tech companies will need to provide regular data to authorities and undergo regular checks by authorities.
Reasons for the Guidelines
The main aim of these guidelines is to curb digital addiction among youth, as government surveys have indicated high levels of digital addiction among children in China. By limiting smartphone usage and introducing age-specific restrictions, the authorities hope to create a healthier digital environment for young users.
Significance of the Proposed Regulations
If adopted, China will introduce some of the strictest regulations worldwide for children's smartphone usage. The guidelines emphasize the government's commitment to protecting children from excessive digital exposure and promoting healthy online habits. By involving both tech companies and parents in enforcing these regulations, the authorities aim to strike a balance between the benefits and potential harms of smartphone usage among children.
GMP System
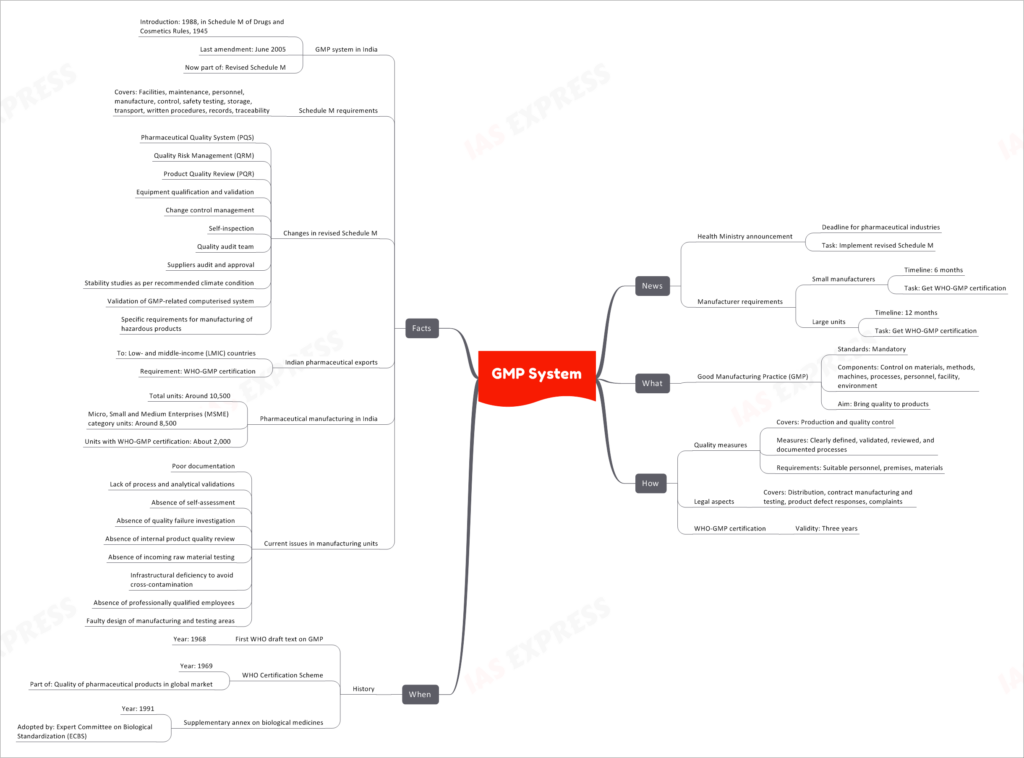
The Health Ministry has announced a deadline for pharmaceutical industries to implement the revised Schedule M, which focuses on Good Manufacturing Practice (GMP) standards. This move aims to bring quality to pharmaceutical products and improve manufacturing practices in India.
What is GMP?
Good Manufacturing Practice (GMP) is a set of mandatory standards that regulate various components of pharmaceutical manufacturing. It ensures control over materials, methods, machines, processes, personnel, facility, and environment, all with the ultimate goal of ensuring the quality of products.
How GMP is Implemented
GMP entails several measures to ensure quality in both production and quality control aspects of pharmaceutical manufacturing. These measures include:
- Clearly defining, validating, reviewing, and documenting processes.
- Meeting suitable personnel, premises, and material requirements.
- Addressing legal aspects such as distribution, contract manufacturing and testing, product defect responses, and complaints.
Pharmaceutical companies seeking compliance with GMP standards are required to obtain WHO-GMP certification, which remains valid for three years.
Historical Development of GMP
The concept of GMP dates back to 1968 when the first WHO draft text on GMP was introduced. The WHO Certification Scheme was established in 1969, and it became part of the broader effort to ensure the quality of pharmaceutical products in the global market. Subsequently, a supplementary annex on biological medicines was adopted in 1991 by the Expert Committee on Biological Standardization (ECBS).
GMP System in India
GMP was introduced in India in 1988 through Schedule M of the Drugs and Cosmetics Rules, 1945. The last amendment to this schedule was made in June 2005, and it is now part of the revised Schedule M.
The revised Schedule M includes several changes to enhance pharmaceutical quality and safety. Some of these changes include:
- Pharmaceutical Quality System (PQS)
- Quality Risk Management (QRM)
- Product Quality Review (PQR)
- Equipment qualification and validation
- Change control management
- Self-inspection
- Quality audit team
- Suppliers audit and approval
- Stability studies as per recommended climate condition
- Validation of GMP-related computerized systems
- Specific requirements for manufacturing hazardous products
Importance of GMP Certification for Indian Pharmaceutical Exports
For Indian pharmaceutical exports to low- and middle-income countries (LMIC), WHO-GMP certification is a crucial requirement. Among the approximately 10,500 pharmaceutical manufacturing units in India, around 8,500 are classified under the Micro, Small, and Medium Enterprises (MSME) category. However, only about 2,000 of these units currently possess WHO-GMP certification.
Current Issues in Manufacturing Units
Several challenges persist in the pharmaceutical manufacturing industry, including:
- Poor documentation
- Lack of process and analytical validations
- Absence of self-assessment and quality failure investigation
- Lack of internal product quality review
- Absence of incoming raw material testing
- Infrastructural deficiencies to avoid cross-contamination
- Lack of professionally qualified employees
- Faulty design of manufacturing and testing areas
Conclusion
GMP is a crucial regulatory framework that ensures the quality and safety of pharmaceutical products. The recent announcement by the Health Ministry to implement the revised Schedule M emphasizes the importance of compliance with GMP standards in India's pharmaceutical industry. By addressing current issues and encouraging adherence to GMP, the country's pharmaceutical sector can strengthen its position in the global market and provide safe and effective medicines to patients worldwide.
e-CARe Portal
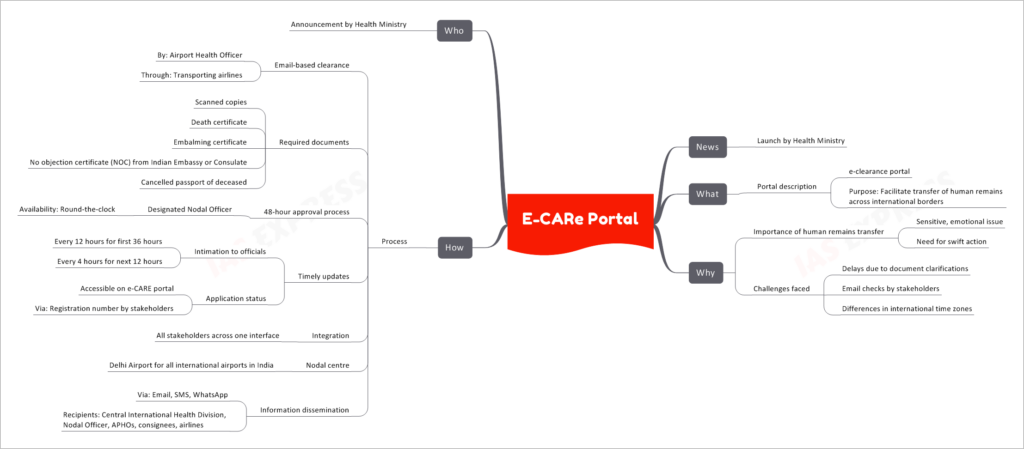
The Health Ministry of India has recently launched the e-CARe portal, a significant step to facilitate the transfer of human remains across international borders. This e-clearance portal aims to address the sensitive and emotional issue of repatriating deceased individuals promptly, streamlining the process and ensuring swift action.
Why the e-CARe Portal is Essential
Transferring human remains across international borders is a critical and sensitive matter. There is a need for quick and efficient procedures to alleviate the emotional burden on grieving families and ensure that the repatriation process proceeds without unnecessary delays.
Challenges Faced in the Past
In the absence of a dedicated portal like e-CARe, the process of repatriation faced several challenges:
- Delays due to Document Clarifications: Verifying and validating the required documents for repatriation often led to unnecessary delays.
- Differences in International Time Zones: Coordinating with officials and airlines across different time zones sometimes caused unavoidable delays.
How the e-CARe Portal Works
The e-CARe portal streamlines the repatriation process by offering an email-based clearance system through the following steps:
1. Email-based Clearance
- Airport Health Officers (APHOs) use the e-CARe portal to initiate the clearance process through the transporting airlines.
2. Required Documents
- The stakeholders are required to submit scanned copies of specific documents, including the death certificate, embalming certificate, no objection certificate (NOC) from the Indian Embassy or Consulate, and the cancelled passport of the deceased.
3. 48-Hour Approval Process
- A designated Nodal Officer, available round-the-clock, ensures a quick 48-hour approval process for the repatriation requests.
4. Timely Updates
- To keep all parties informed, officials receive intimation every 12 hours for the first 36 hours and every 4 hours for the next 12 hours.
5. Application Status Accessibility
- Stakeholders can track the application status through the e-CARe portal using the provided registration number.
6. Integration and Nodal Centre
- The e-CARe portal brings together all stakeholders on one platform, ensuring seamless communication. The Delhi Airport serves as the nodal centre for all international airports in India.
7. Information Dissemination
- The e-CARe portal disseminates information via email, SMS, and WhatsApp to various recipients, including the Central International Health Division, Nodal Officer, APHOs, consignees, and airlines.
Conclusion
The launch of the e-CARe portal by the Health Ministry marks a significant milestone in the process of repatriating human remains across international borders. By streamlining the clearance process and providing timely updates through a dedicated platform, the e-CARe portal aims to ease the burden on grieving families and ensure that the transfer of human remains is handled with sensitivity and efficiency. This initiative reflects the government's commitment to addressing the challenges faced in the past and ensuring a smoother and more compassionate repatriation process for all.
Import Restrictions on Laptops & Computers
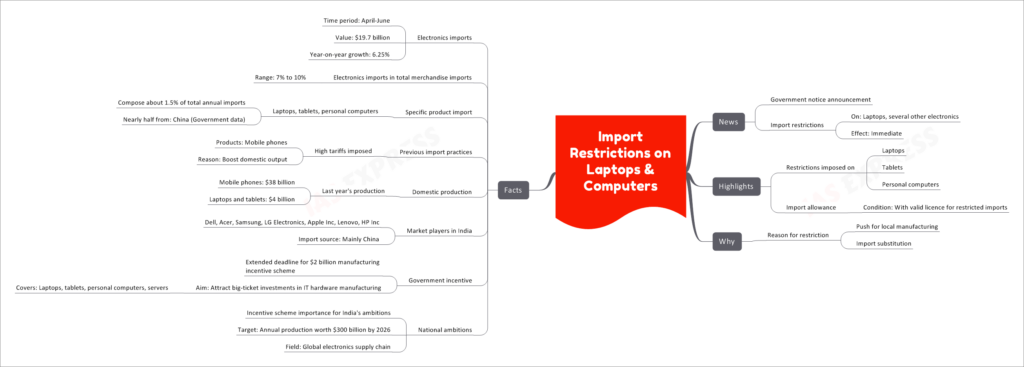
In recent news, the government of India has announced immediate import restrictions on laptops, tablets, and personal computers. This move comes as a part of the country's push for promoting local manufacturing and import substitution in the electronics sector.
Highlights of the Import Restrictions
The import restrictions apply to the following products:
- Laptops
- Tablets
- Personal computers
Importing these electronics will now require a valid license for restricted imports, making it more challenging for foreign manufacturers to enter the Indian market.
Reasons for the Restrictions
The government has imposed these import restrictions with two primary objectives:
- Push for Local Manufacturing: By restricting imports, the government aims to encourage domestic production of laptops and computers. This move is part of a broader effort to boost the country's manufacturing capabilities and reduce dependence on imports.
- Import Substitution: Restricting the import of laptops and computers is an attempt to promote domestic alternatives and encourage consumers to buy locally manufactured products instead of foreign ones.
Facts and Figures
Electronics Imports
- During the period from April to June, electronics imports amounted to $19.7 billion, with a year-on-year growth of 6.25%.
- Electronics imports make up around 7% to 10% of India's total merchandise imports.
- Laptops, tablets, and personal computers together constitute about 1.5% of the country's total annual imports in the electronics sector.
- Almost half of these imports come from China, as per government data.
Previous Import Practices
- The government previously imposed high tariffs on mobile phones to promote domestic output and reduce reliance on imports.
Domestic Production
- In the previous year, India's domestic production of mobile phones reached $38 billion, while laptops and tablets manufacturing amounted to $4 billion.
Market Players and Import Source
- Major market players in India's electronics sector include Dell, Acer, Samsung, LG Electronics, Apple Inc, Lenovo, and HP Inc.
- The primary import source for these electronics products is China.
Government Incentive
- The government has extended the deadline for a $2 billion manufacturing incentive scheme aimed at attracting big-ticket investments in IT hardware manufacturing, including laptops, tablets, personal computers, and servers.
National Ambitions
- The incentive scheme holds significant importance for India's ambition of achieving annual production worth $300 billion by 2026 in the global electronics supply chain.
Conclusion
The import restrictions on laptops and computers reflect the Indian government's commitment to boost local manufacturing and promote domestic production in the electronics sector. By limiting imports and encouraging investments through incentive schemes, India aims to strengthen its position in the global electronics market and reduce its dependence on foreign imports. As the country moves forward with its ambitious target for domestic production, it is likely to witness significant changes in the electronics landscape and contribute to the growth of the Indian economy.
Polar Research Vessel
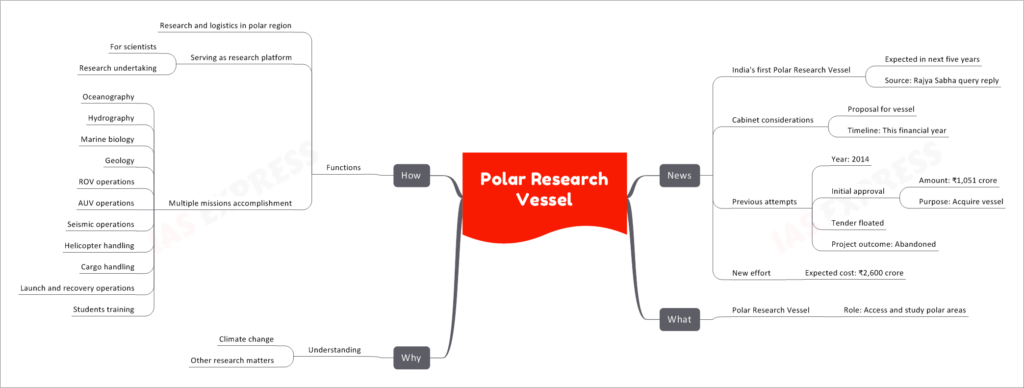
India's first Polar Research Vessel is expected to be ready within the next five years, marking a significant step forward for the nation's scientific exploration capabilities.
India's First Polar Research Vessel
The Polar Research Vessel is a specialized ship designed to access and study the polar regions. It will serve as a critical tool for scientists to gain deeper insights into various aspects of polar environments and contribute to important research on climate change and other related matters.
Previous Attempts and the New Effort
India's journey towards acquiring a Polar Research Vessel began back in 2014 when an initial approval was granted, allocating ₹1,051 crore for the purpose. However, the project faced challenges and was eventually abandoned after floating a tender. Undeterred, the country is now making a renewed effort with an expected budget of ₹2,600 crore to bring this ambitious project to fruition.
The Importance of a Polar Research Vessel
Understanding Climate Change
The polar regions play a crucial role in the global climate system. The research vessel will enable scientists to gather data on melting ice, temperature changes, and ocean currents, leading to a better understanding of climate change and its impact on the planet.
Exploring Other Research Matters
Beyond climate change, polar regions are home to diverse ecosystems and unique geological features. The vessel will facilitate research in fields such as oceanography, hydrography, marine biology, geology, and more, contributing to broader scientific knowledge.
Functions of the Polar Research Vessel
The Polar Research Vessel will be equipped with a wide range of capabilities, making it a versatile research platform.
Research and Logistics in Polar Regions
One of the primary functions of the vessel is to support scientific research missions in the harsh and challenging polar environments. It will serve as a mobile laboratory, allowing scientists to conduct experiments and collect samples directly from the polar regions.
Accommodating Scientists and Research Undertakings
The vessel will provide state-of-the-art facilities and accommodations for scientists on board, ensuring they can perform their research tasks effectively. It will serve as a hub for various research undertakings and projects, fostering collaboration among scientists from different disciplines.
Accomplishing Multiple Missions
The Polar Research Vessel will be capable of executing various missions, including:
- Oceanography: Studying the ocean and its physical properties, currents, and marine life.
- Hydrography: Mapping and studying the underwater topography and features.
- Marine Biology: Investigating marine ecosystems and their inhabitants.
- Geology: Studying the geological formations and processes in polar regions.
- ROV Operations: Deploying remotely operated vehicles for underwater exploration.
- AUV Operations: Utilizing autonomous underwater vehicles for data collection.
- Seismic Operations: Conducting seismic surveys to understand Earth's structure.
- Helicopter Handling: Enabling access to remote locations via helicopters.
- Cargo Handling: Transporting equipment and supplies for research missions.
- Launch and Recovery Operations: Deploying and retrieving research equipment.
- Students Training: Providing opportunities for aspiring researchers to gain hands-on experience.