Controlled Human Infection Studies
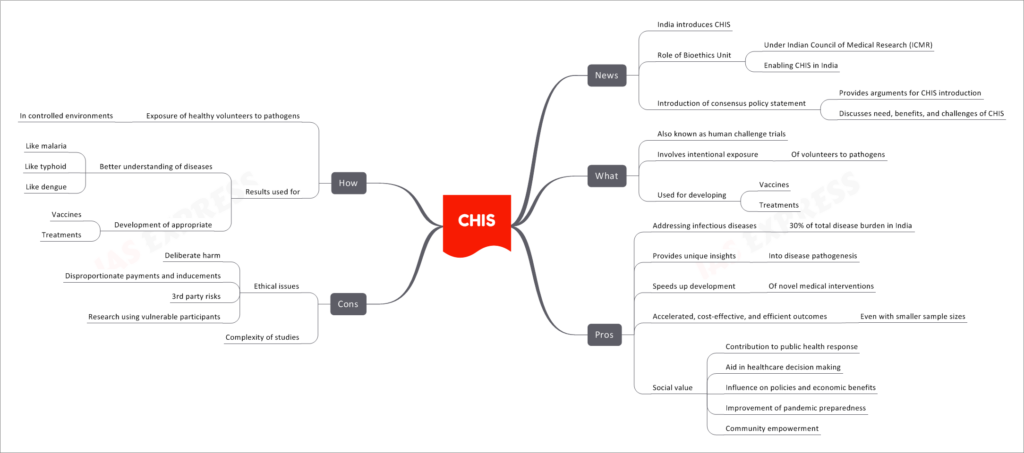
Controlled Human Infection Studies (CHIS), also known as human challenge trials, have been introduced in India. The Bioethics Unit under the Indian Council of Medical Research (ICMR) plays a crucial role in enabling CHIS in the country. A consensus policy statement has been introduced to provide arguments for CHIS’s introduction, discussing the need, benefits, and challenges associated with such studies.
This topic of “Controlled Human Infection Studies” is important from the perspective of the UPSC IAS Examination, which falls under General Studies Portion.
What
- Controlled Human Infection Studies involve intentional exposure of volunteers to pathogens. These studies are primarily used for the development of vaccines and treatments.
Pros
- Addressing Infectious Diseases: CHIS can help in tackling infectious diseases that contribute to 30% of the total disease burden in India.
- Unique Insights: These studies provide researchers with unique insights into disease pathogenesis.
- Speeding up Development: CHIS accelerates the development of novel medical interventions.
- Cost-Effective and Efficient Outcomes: CHIS can lead to accelerated, cost-effective, and efficient outcomes, even with smaller sample sizes.
- Social Value: The studies offer various social benefits, including contributing to public health responses, aiding healthcare decision-making, influencing policies and economic benefits, improving pandemic preparedness, and empowering communities.
Cons
- Ethical Issues: CHIS raises ethical concerns due to deliberate harm caused to volunteers, the possibility of disproportionate payments and inducements, risks to third parties, and the involvement of vulnerable participants in research.
- Complexity of Studies: Conducting CHIS can be complex and requires careful planning and ethical considerations.
How
- CHIS involves exposing healthy volunteers to pathogens in controlled environments.
- The results obtained from these studies are used to better understand diseases like malaria, typhoid, and dengue, and to develop appropriate vaccines and treatments.
Conclusion
Controlled Human Infection Studies (CHIS) present a potential pathway for addressing infectious diseases and accelerating the development of medical interventions in India. While these studies offer valuable insights and social benefits, they also come with ethical challenges that must be carefully addressed. With the involvement of the Bioethics Unit under the Indian Council of Medical Research, the introduction of CHIS in India is expected to be guided by a thoughtful and responsible approach.

