Chemistry Nobel 2023
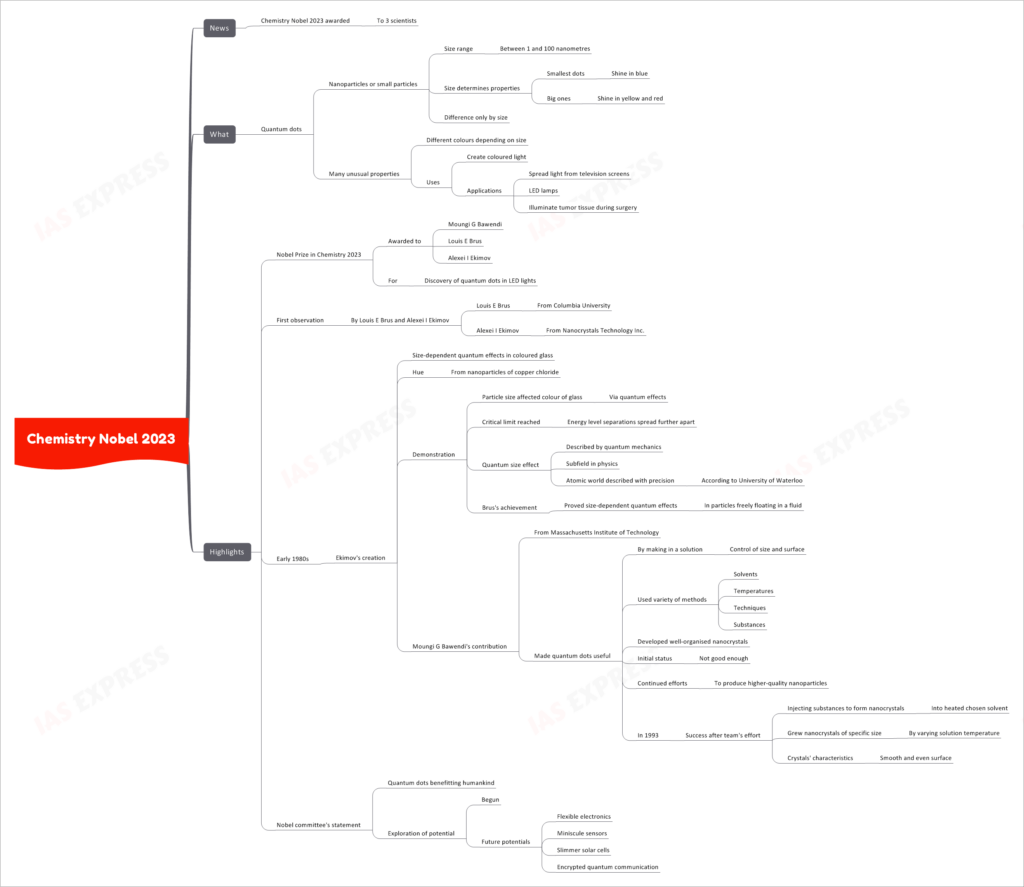
In a recent groundbreaking announcement, the Nobel Prize in Chemistry for 2023 has been awarded to three distinguished scientists for their pioneering work in the realm of quantum dots. This prestigious recognition marks a significant milestone in the field of nanotechnology and its transformative impact on various applications, from LED lights to medical advancements. Let’s delve into the details of this remarkable discovery.
This topic of “Chemistry Nobel 2023” is important from the perspective of the UPSC IAS Examination, which falls under General Studies Portion.
What Are Quantum Dots?
Nanoparticles of Remarkable Potential
Quantum dots, at their core, are nanoparticles or incredibly tiny particles with sizes that fall within the range of 1 to 100 nanometres. It’s important to note that the size of these quantum dots plays a pivotal role in determining their unique properties. These properties can vary significantly depending on their size, making quantum dots a versatile tool with a multitude of applications.
Size Dictates Their Properties
The most remarkable feature of quantum dots is how their size directly influences their behavior:
- Smallest Dots: Quantum dots at the lower end of the size spectrum tend to emit a brilliant blue light when stimulated.
- Larger Ones: As the size of quantum dots increases, their emitted light shifts towards the yellow and red spectrum.
Unusual Properties Abound
Quantum dots are not just limited to their size-dependent color variations. They exhibit numerous other exceptional characteristics, including:
- Diverse Colors: Quantum dots can produce a wide range of colors depending on their size, making them invaluable for creating vibrant displays and lighting.
- Applications Galore: These tiny wonders find use in an array of applications, such as enhancing the brightness and color accuracy of television screens, improving the efficiency of LED lamps, and even illuminating tumor tissue during delicate surgical procedures.
Highlights of the Nobel Prize in Chemistry 2023
The Laureates
This year’s Nobel Prize in Chemistry is a tribute to the groundbreaking work of three exceptional scientists:
- Moungi G. Bawendi: Hailing from the Massachusetts Institute of Technology, Bawendi made quantum dots practical by developing methods to control their size and surface properties.
- Louis E. Brus: A researcher at Columbia University, Brus contributed to the understanding of size-dependent quantum effects in particles freely suspended in a fluid.
- Alexei I. Ekimov: Ekimov, affiliated with Nanocrystals Technology Inc., was one of the early pioneers who observed size-dependent quantum effects in colored glass back in the early 1980s.
The Genesis of Quantum Dots
- Louis E. Brus and Alexei I. Ekimov: The journey into the world of quantum dots began with Louis E. Brus and Alexei I. Ekimov. Ekimov’s work in the early 1980s involved creating size-dependent quantum effects in colored glass, using nanoparticles of copper chloride. This work demonstrated how the size of particles directly impacted the color of glass, showcasing the role of quantum effects.
- Critical Quantum Size Effect: Brus took Ekimov’s discoveries a step further by proving that size-dependent quantum effects weren’t limited to glass; they applied to particles freely suspended in a fluid, as well. These breakthroughs provided a profound insight into the atomic world described with precision by quantum mechanics, a subfield in physics.
Moungi G. Bawendi’s Contribution
- Harnessing Quantum Dots: Moungi G. Bawendi’s contributions from MIT were instrumental in making quantum dots practical for a wide range of applications. He achieved this by developing methods to create quantum dots in a solution, allowing for precise control of their size and surface characteristics.
- Persistent Efforts: Bawendi’s journey was not without challenges. Initially, the quality of nanocrystals produced was subpar. However, he and his team persisted in their efforts, leading to a breakthrough in 1993. They successfully grew nanocrystals of specific sizes by injecting substances into a carefully chosen, heated solvent. These nanocrystals exhibited smooth and even surfaces, paving the way for countless innovations.
Quantum Dots: A Benefit to Humankind
The Nobel committee’s statement underscores the profound impact of quantum dots on humankind. This discovery has not only opened up new possibilities but has also paved the way for the exploration of untapped potential.
The Journey Begins
The journey of quantum dots began with the first observations of size-dependent quantum effects by Louis E. Brus and Alexei I. Ekimov in the early 1980s. These early breakthroughs set the stage for a transformative journey into the world of nanotechnology.
Future Potential
As we look to the future, the potential applications of quantum dots appear boundless. Some exciting prospects include the development of flexible electronics, minuscule sensors, more efficient and compact solar cells, and even encrypted quantum communication methods.

