Nuclear Weapons
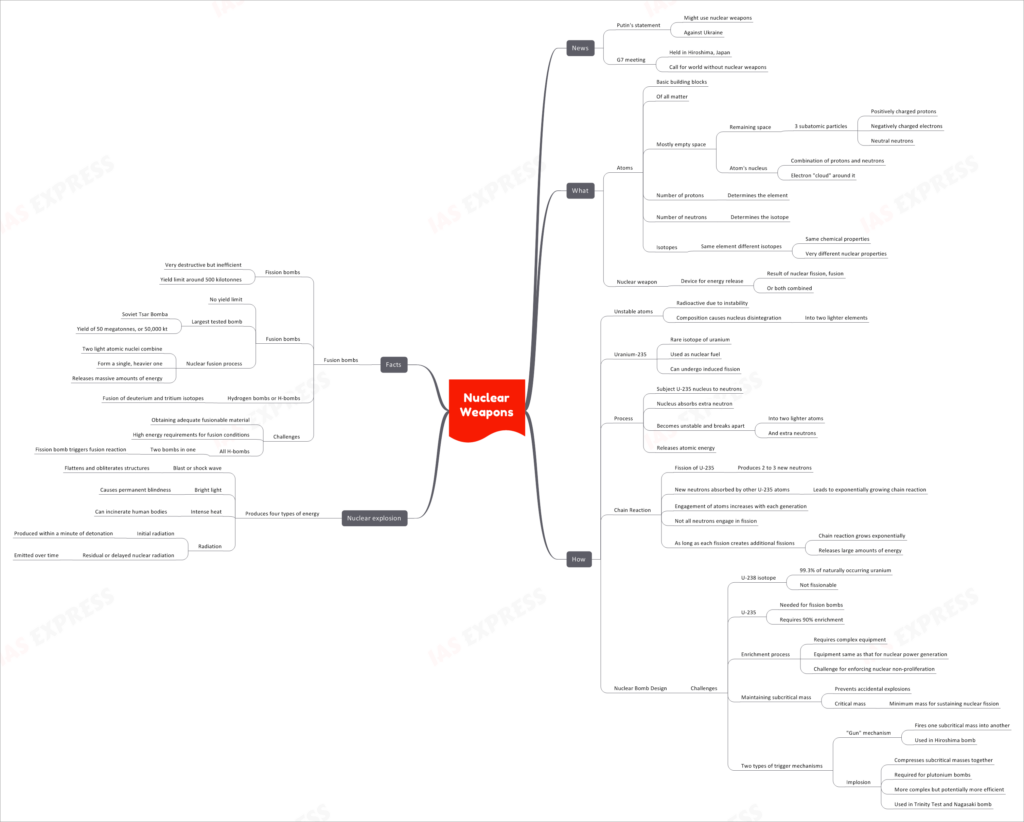
Recent news highlights the growing concerns about nuclear weapons, particularly in the context of geopolitical tensions. Putin’s statement about using nuclear weapons against Ukraine and the G7 meeting in Hiroshima, Japan, calling for a world without nuclear weapons, have brought the issue to the forefront.
This topic of “Nuclear Weapons” is important from the perspective of the UPSC IAS Examination, which falls under General Studies Portion.
What are Atoms?
Atoms are the fundamental building blocks of all matter, consisting of mostly empty space. They contain three subatomic particles: positively charged protons, negatively charged electrons, and neutral neutrons. The atom’s nucleus is a combination of protons and neutrons, with an electron “cloud” surrounding it. The number of protons determines the element, while the number of neutrons determines the isotope.
Understanding Nuclear Weapons
Nuclear weapons are devices designed to release tremendous amounts of energy through nuclear fission, fusion, or a combination of both.
How Unstable Atoms Lead to Nuclear Fission
Unstable atoms, often referred to as radioactive isotopes, undergo a process called nuclear fission. During fission, the nucleus of an unstable atom disintegrates into two lighter elements, along with the release of extra neutrons. Uranium-235 (U-235) is a rare isotope of uranium used as nuclear fuel because it can undergo induced fission.
The Process of Nuclear Fission
In a nuclear bomb, U-235 nuclei are subjected to neutrons. When a nucleus absorbs an extra neutron, it becomes unstable and breaks apart into two lighter atoms and releases additional neutrons. This process results in the release of atomic energy.
Chain Reaction: Amplifying the Power
The fission of U-235 produces 2 to 3 new neutrons, which are absorbed by other U-235 atoms, leading to an exponentially growing chain reaction. As more atoms engage in fission with each generation, the chain reaction releases large amounts of energy.
Design Challenges for Nuclear Bombs
Creating a nuclear bomb is a complex task, with several challenges to overcome:
- Enrichment Process: The isotope U-238, comprising 99.3% of naturally occurring uranium, is not fissionable. To build fission bombs, U-235 requires 90% enrichment, which demands sophisticated equipment similar to that used for nuclear power generation.
- Maintaining Subcritical Mass: Preventing accidental explosions involves keeping the mass of nuclear material below the critical mass, the minimum mass required for sustaining nuclear fission.
- Trigger Mechanisms: Nuclear bombs use two types of trigger mechanisms: the “gun” mechanism, where one subcritical mass is fired into another (used in Hiroshima bomb), and implosion, which compresses subcritical masses together (required for plutonium bombs).
The Nuclear Explosion
A nuclear explosion releases four types of energy:
- Blast or Shock Wave: Flattens and obliterates structures in the vicinity of the explosion.
- Bright Light: Can cause permanent blindness for those exposed to it.
- Intense Heat: Can incinerate human bodies and objects nearby.
- Radiation: Initial radiation is produced within a minute of detonation, while residual or delayed nuclear radiation is emitted over time, posing long-term health hazards.
Facts about Nuclear Bombs
Fusion Bombs vs. Fission Bombs
- Fission Bombs: Although very destructive, they are inefficient, with a yield limit around 500 kilotonnes.
- Fusion Bombs: Also known as hydrogen bombs or H-bombs, have no yield limit. The largest tested bomb was the Soviet Tsar Bomba, with a yield of 50 megatonnes, or 50,000 kilotonnes.
Nuclear Fusion Process
In fusion bombs, two light atomic nuclei combine to form a single, heavier one, releasing massive amounts of energy in the process.
Challenges with Fusion Bombs
Creating fusion bombs poses several challenges, including obtaining adequate fusionable material and the high energy requirements to achieve fusion conditions. All H-bombs have two bombs in one, where a fission bomb triggers the fusion reaction.
